More Articles
Withings, the healthcare technology company known for its remote monitoring devices, announced that the U.S. Food and Drug Administration (FDA) has cleared its newest smart scale, the Body Scan…
ESC Congress 2023, the annual meeting of the European Society of Cardiology (ESC), is scheduled for Aug. 25-28 in Amsterdam.
ESC Congress is often described as…
Albert L. Waldo, MD, an influential cardiologist who helped shape the field of electrophysiology, died Aug. 17 due to complications related to a long fight…
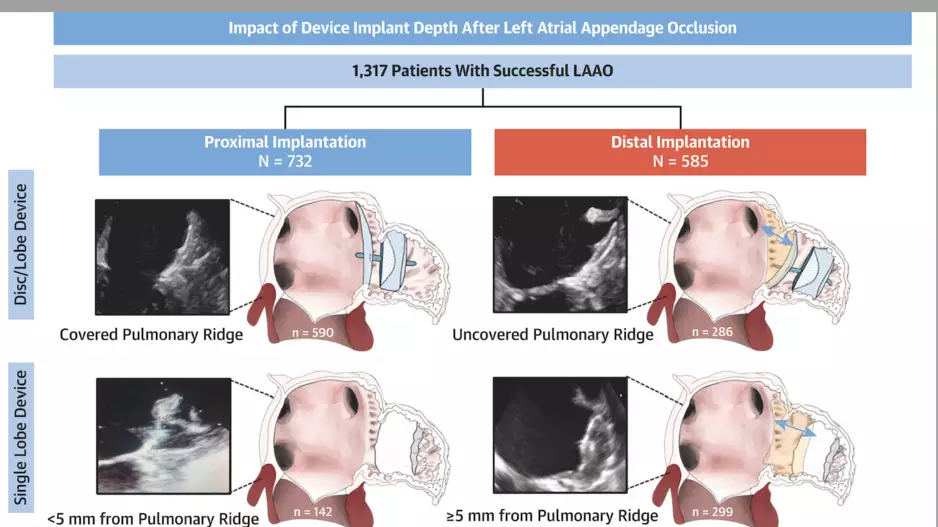
Deeper device implantation during left atrial appendage occlusion (LAAO) procedures increases the patient’s risk of developing a blood clot, according to new research published in JACC: Cardiovascular Interventions.[1]…
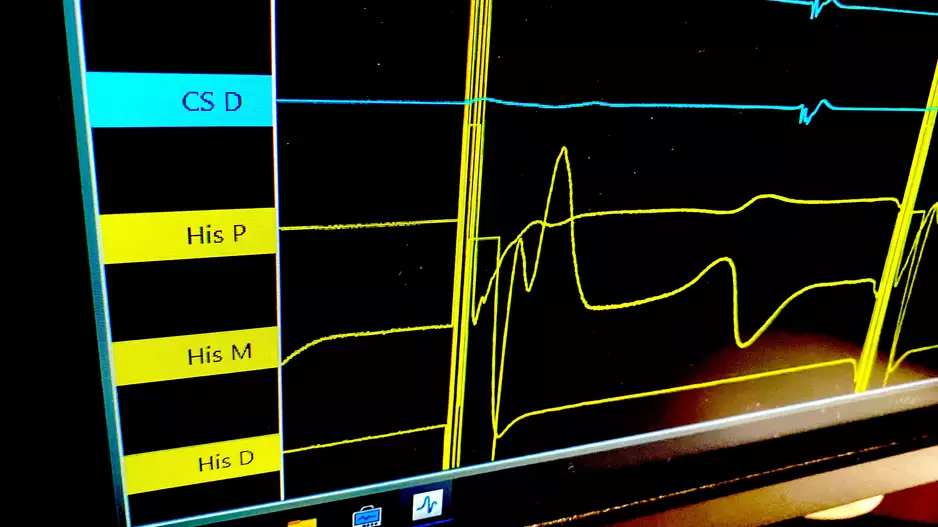
CathVision, a Denmark-based healthcare technology company focused on the electrophysiology lab, has received U.S. Food and Drug…
Viz.ai has received a de novo approval from the U.S. Food…
The U.S. Food and Drug Administration (FDA) has issued a new draft guidance designed to help applicants add clinically relevant information about QTc interval prolongation to the labeling of non-…
Can fasting instructions before transcatheter aortic valve replacement (TAVR), ablation and other cardiac procedures be relaxed? A team of…
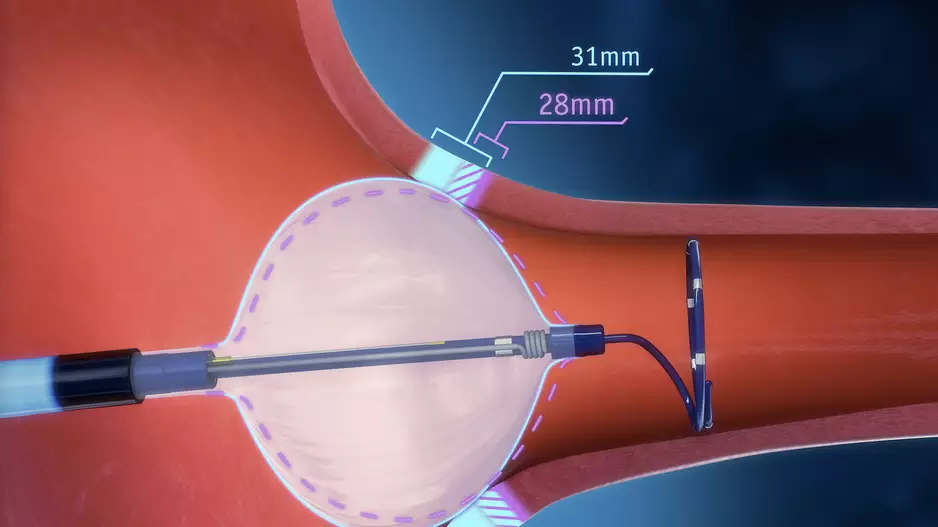
The U.S. Food and Drug Administration (FDA) has approved Boston Scientific’s POLARx Cryoablation System for the treatment of patients with atrial fibrillation (AFib).
…
NFL player feeling thankful after AFib diagnosis: ‘I think it’s just the good Lord looking after me’
Tyler Shatley, a veteran offensive lineman with the Jacksonville Jaguars, was diagnosed with atrial fibrillation (AFib) on Aug. 2 after experiencing symptoms for the last few years. Shatley discussed the diagnosis with the media on…
Biosense Webster, a Johnson & Johnson company, has received U.S. Food and Drug Administration (FDA) approval for multiple devices from its cardiac ablation portfolio to be used as part of a zero-…
Expanding Medicaid eligibility is associated with significant improvements in care for low-income patients with coronary artery disease, heart failure,…
The U.S. Food and Drug Administration (FDA) has announced that GE Healthcare is recalling more than 7,000 TruSignal SpO2 sensors due to issues with the devices malfunctioning and sending a reduced…
Patients who develop atrial fibrillation (AFib) for the first time after transcatheter aortic valve replacement (TAVR) face a heightened…
PaceMate, the Florida-based healthcare technology company focused on remote cardiac monitoring, has received a new “strategic growth investment” from Lead Edge Capital, a well-known tech investor that has previously worked with such…
Targeting immune cells that play a key role in the development of atrial fibrillation (AFib) could give cardiologists a new way to treat the disease, according to a new analysis in Science.[1] The study’s authors said this…
The U.S. Food and Drug Administration (FDA) has announced that Medtronic is recalling nearly 350,000 implantable cardiac devices due to continued issues with their ability to deliver high voltage…
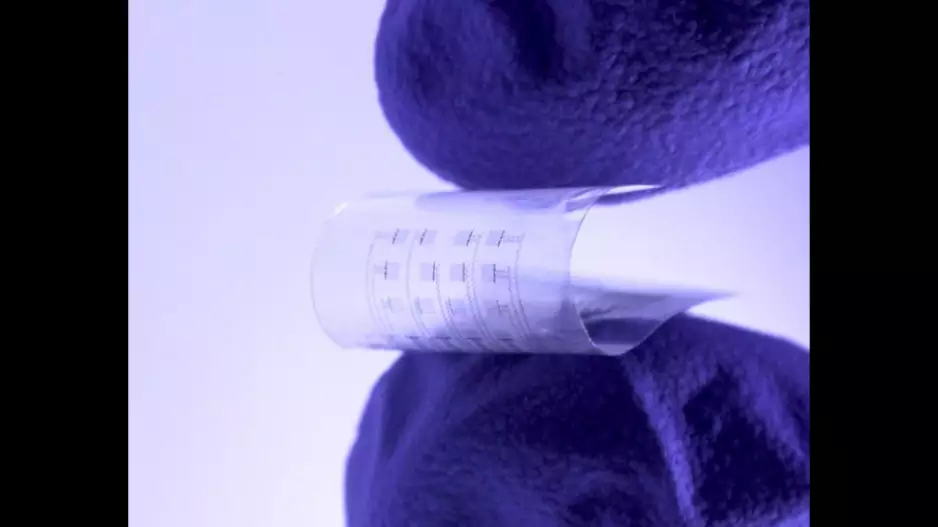
Researchers have developed new heart monitors that dissolve inside the body when they are no longer needed, sharing their work in Science Advances.[1]
The new-look devices are soft, flexible…
The U.S. Food and Drug Administration (FDA) has approved Abbott’s Aveir dual chamber (DR) leadless pacemaker system, the world’s very first dual-chamber leadless pacing solution for treating patients…
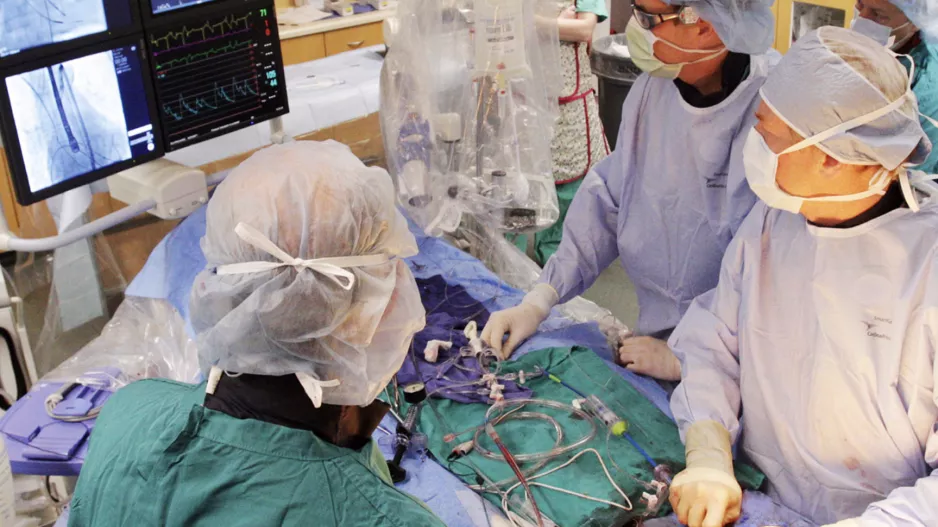
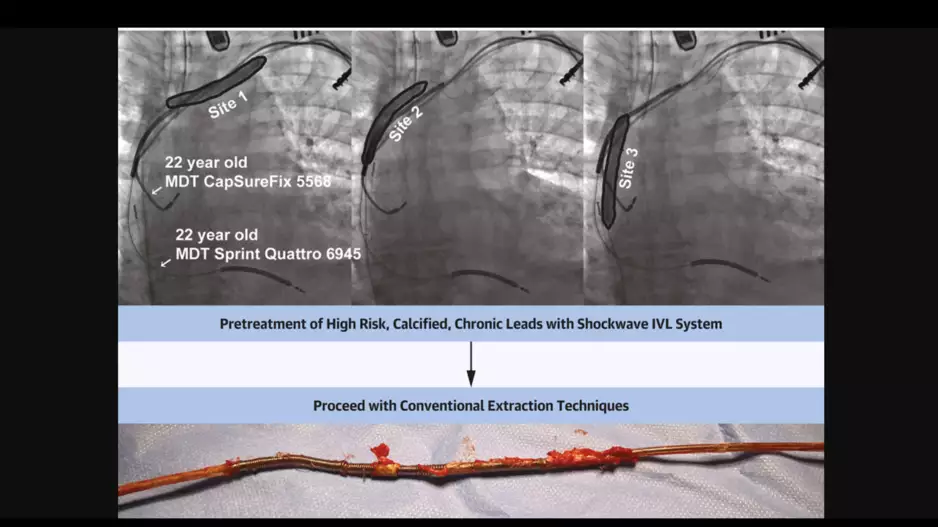
Intravascular lithotripsy (IVL) can provide significant value during transvenous lead extraction (TLE) procedures by identifying dense calcifications, according to new research published in JACC: Clinical Electrophysiology.[1…
Adding electrocardiogram (ECG) sensors to shopping carts can helps identify patients who may have atrial fibrillation (AFib) or other…