More Articles
The U.S. Food and Drug Administration (FDA) has announced that Medtronic is recalling 87,709 implantable cardiac devices due to the risk of a malfunction that could result in serious injury or death…
CathVision, a Denmark-based healthcare technology company focused on the electrophysiology (EP) lab, has closed a new financing round worth $7.2 million.…
Monitoring patient symptoms is an essential part of diagnosing and treating cardiovascular disease (CVD). A new scientific statement from the American Heart Association (AHA), published in …
Atrial cardiomyopathy is associated with an increased risk of dementia among older adults, according to new research published in the Journal of the American Heart Association.[1]
“Stroke…
In a massive simulation exercise, researchers recently found that using wrist-worn wearable devices to screen people for atrial fibrillation (AFib) may be more cost-effective than traditional modalities such as pulse palpation and 12-…
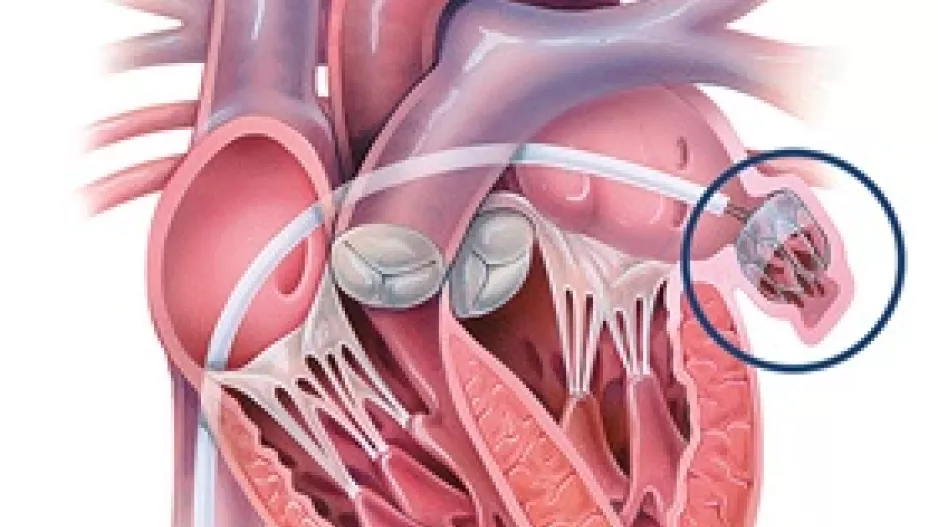
The risk of in-hospital mortality may be significantly higher among transcatheter edge-to-edge repair (TEER) patients who present with concomitant…
There is not agreement among some of the top structural heart experts who perform left atrial appendage occlusion (LAAO) if cardiac computed tomography (CT…
The U.S. Food and Drug Administration (FDA) has had a busy in July, approving…
Patients with heart failure or…
The KardiaBand device from AliveCor is a more reliable tool for identifying atrial fibrillation (AFib) than the…
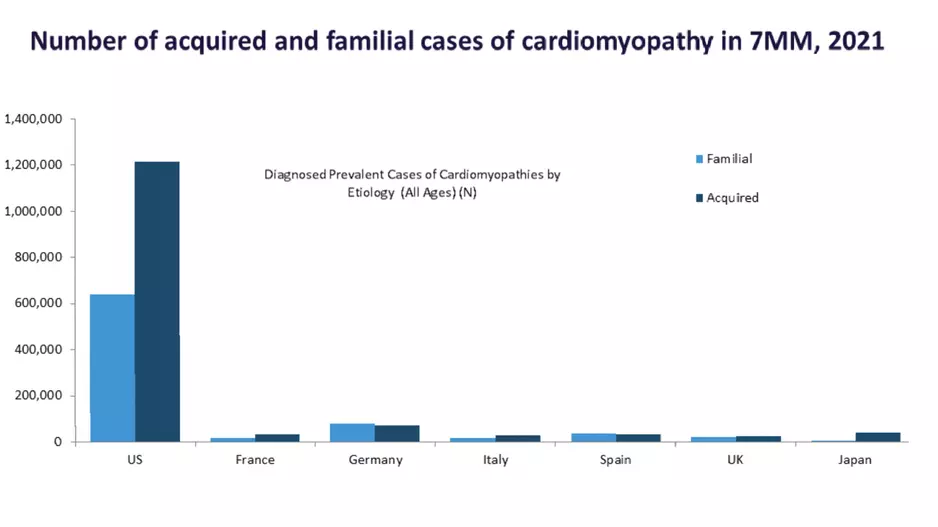
A new healthcare market report projects the United States will see a major increase in new cardiomyopathy cases in coming years, possibly reaching more than 2.3 million by 2031. The authors of the report said hospitals should consider the…
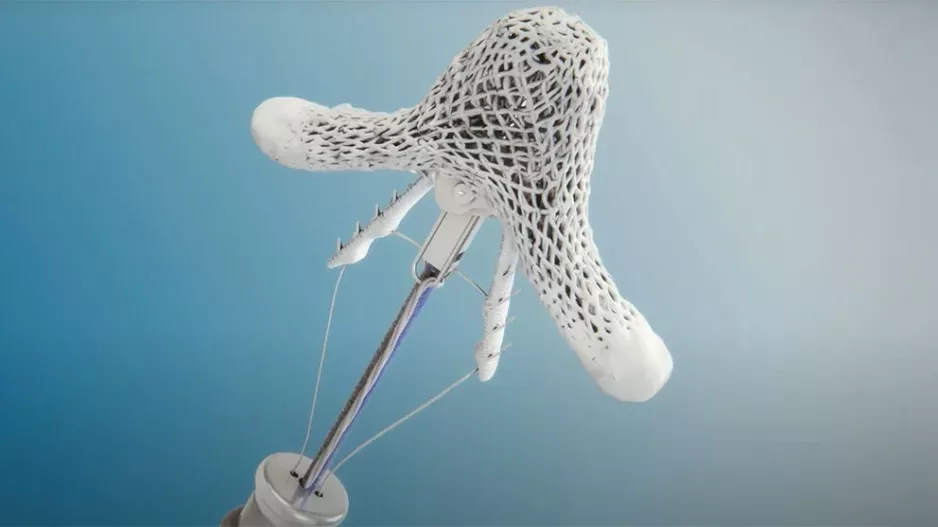
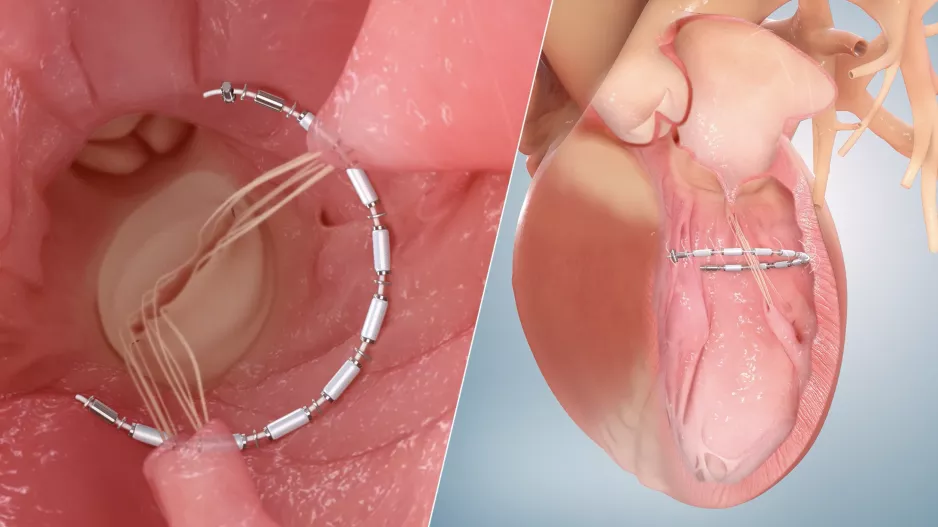
There are now two transcatheter left atrial appendage (LAA) occlusion devices cleared by the U.S. Food and Drug Administration (FDA) and a couple more are expected to soon enter FDA investigational device exemption (IDE) trials. Experts…
Patients with a history of atrial fibrillation (AFib) are more likely to experience adverse outcomes within two years of transcatheter…
When the first catheter-implanted, leadless pacemaker was introduced in 2016, there was a lot of interest in this minimally invasive technology. But the biggest criticism was that it only addressed the needs of about 20% of patients. However, 100…
Catheter ablation is an "economically attractive" treatment option for atrial fibrillation (AFib) when compared to antiarrhythmic drug therapy alone,, according to a new study in …
Patients who develop atrial fibrillation (AFib) after surgery face a higher risk of being hospitalized for heart failure, according to new data published in the European Heart Journal.[1]
The…
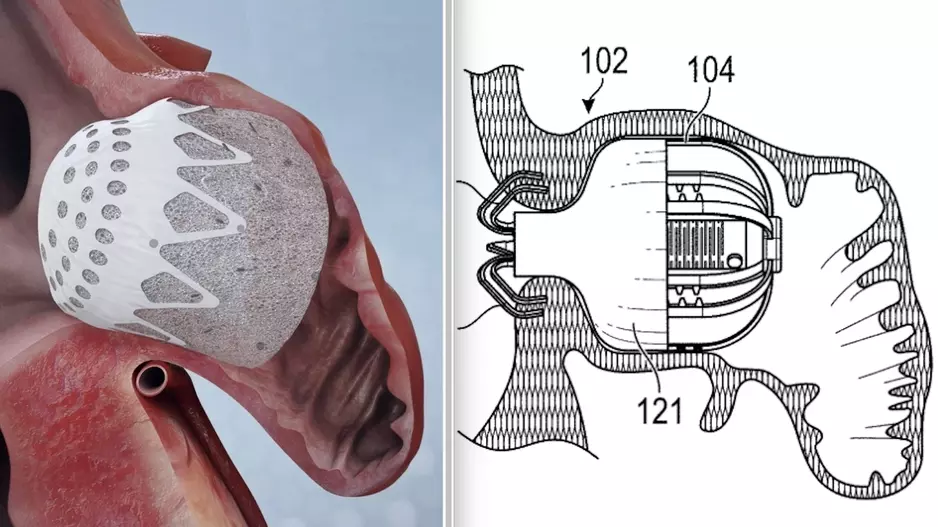
Large peridevice leaks after left atrial appendage occlusion (LAAO) are incredibly rare and not associated with a greater risk of adverse outcomes, according to new research published in JACC: Clinical Electrophysiology.[1]…
Marijuana use among older adults is associated with a heightened risk of atrial and ventricular arrhythmias, according to new research published in the American Journal of Cardiology.[1]
“…
Non-vitamin K antagonist oral anticoagulants (NOACs) may be a safe alternative to warfarin when treating patients with atrial fibrillation (AFib) and valvular heart disease (VHD), according to new research published in the …
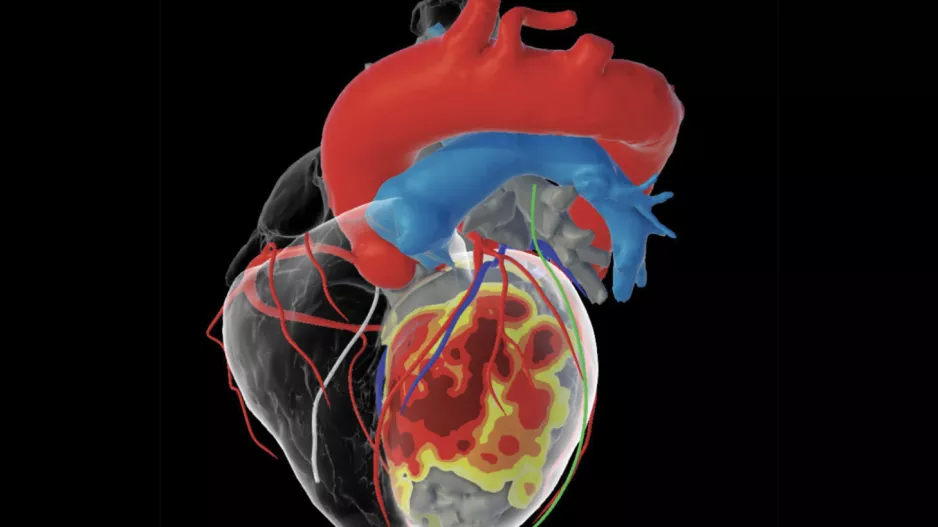
inHEART Medical, an international healthcare company with offices in France and the United States, has received clearance from the U.S. Food and Drug Administration (FDA) for its new 3D visualization…




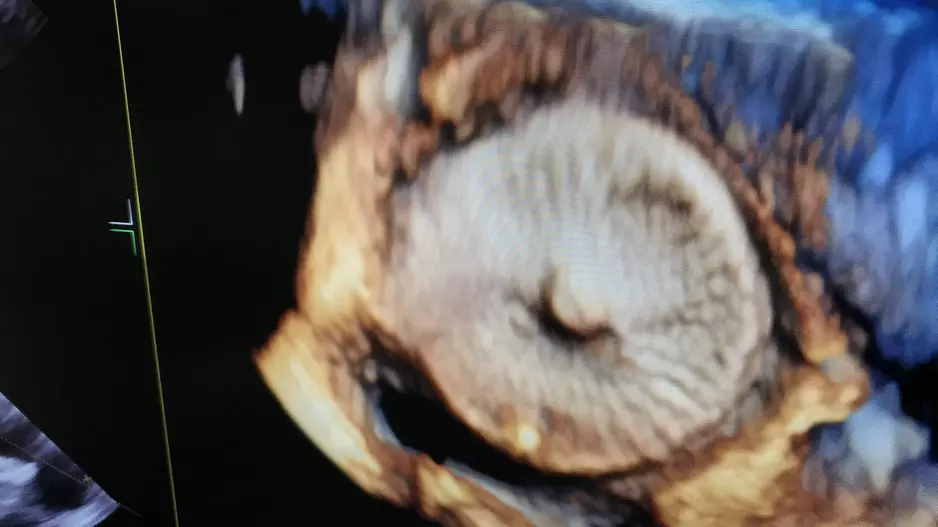
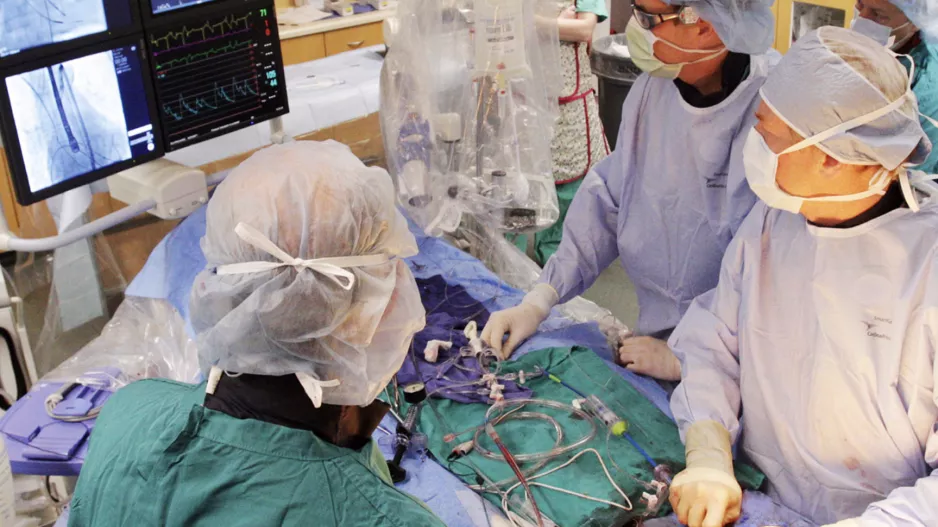
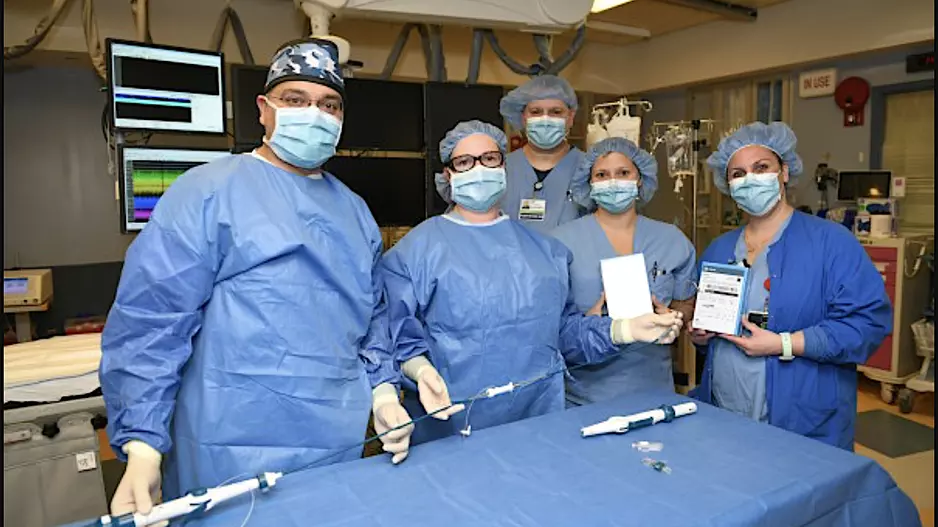

![Large peridevice leaks after left atrial appendage occlusion (LAAO) are incredibly rare and not associated with a greater risk of adverse outcomes, according to new research published in JACC: Clinical Electrophysiology.[1] Smaller residual links are more common, however, and associated with a risk of thromboembolic and bleeding events.](/sites/default/files/styles/media_image/public/2018-08/olderpatient-doctor.jpg.webp?itok=WkwdpD4_)