More Articles
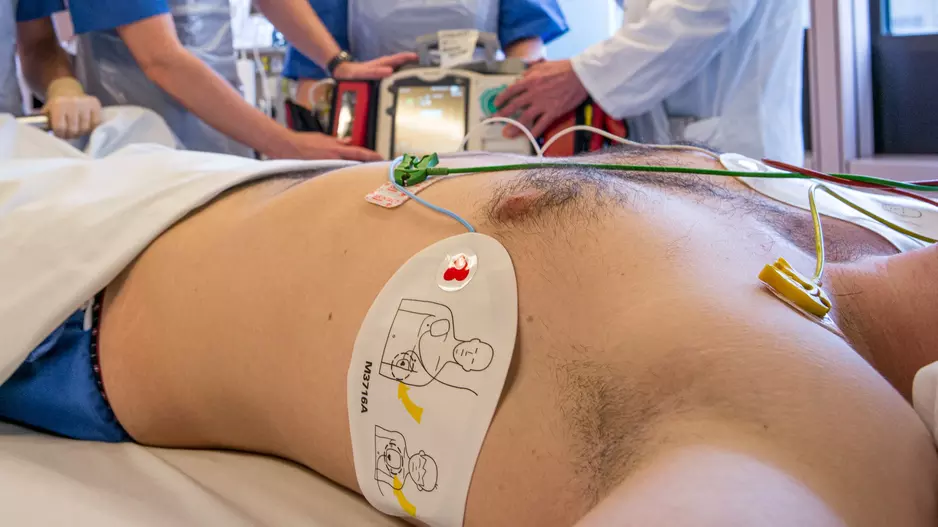
Marijuana use among older adults is associated with a heightened risk of atrial and ventricular arrhythmias, according to new research published in the American Journal of Cardiology.[1]
“…
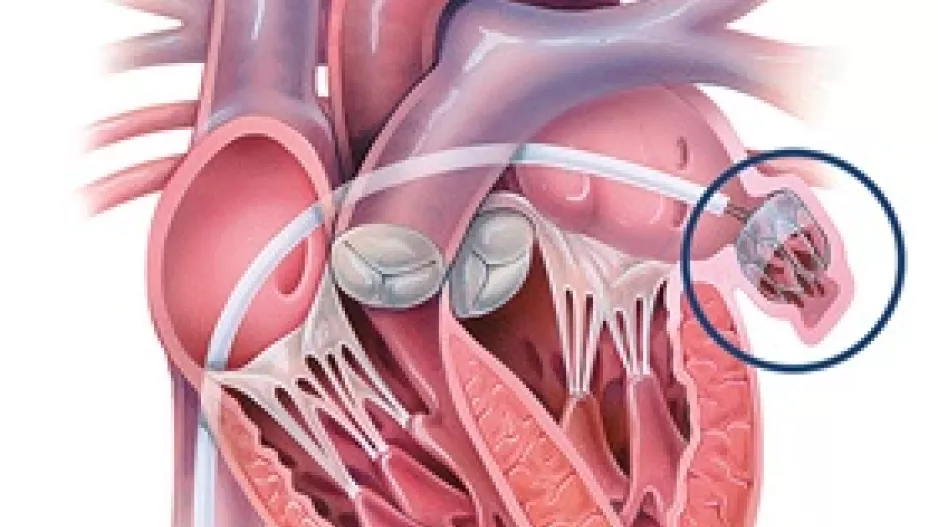
Non-vitamin K antagonist oral anticoagulants (NOACs) may be a safe alternative to warfarin when treating patients with atrial fibrillation (AFib) and valvular heart disease (VHD), according to new research published in the …
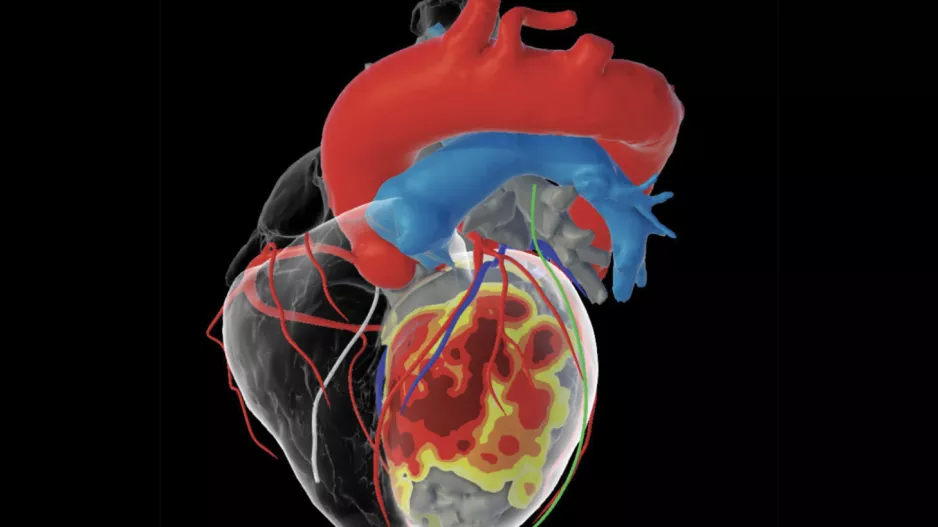
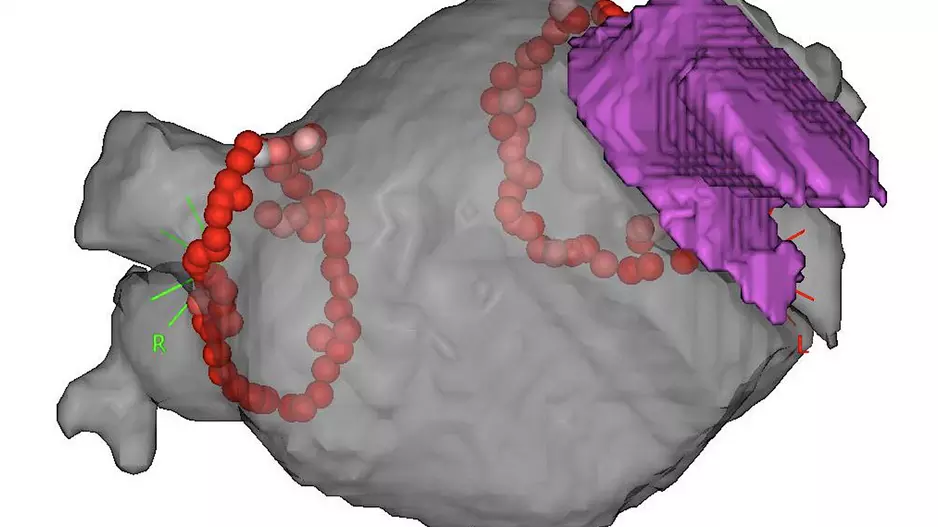
inHEART Medical, an international healthcare company with offices in France and the United States, has received clearance from the U.S. Food and Drug Administration (FDA) for its new 3D visualization…
Left bundle branch block (LBBB), one of the most common complications after transcatheter aortic valve replacement (TAVR), is…
It has been a busy few weeks for the U.S. Food and Drug Administration (FDA). The agency…
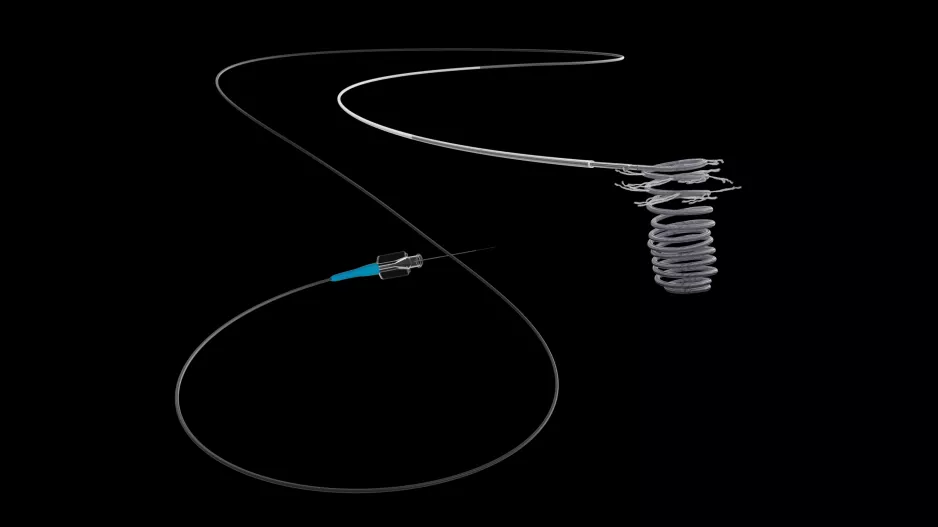
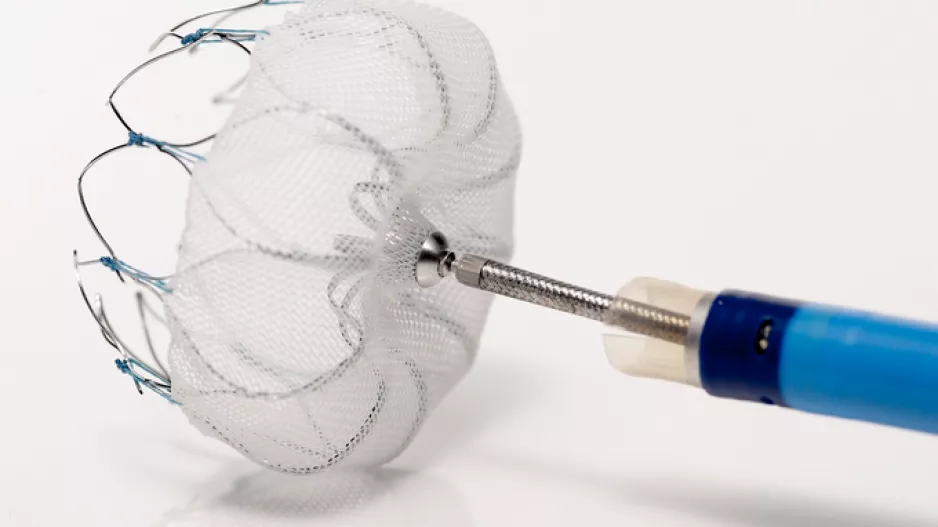
Atrial fibrillation patients who underwent left atrial appendage occlusion (LAAO) with the Watchman device after it gained U.S. FDA approval have experienced fewer complications and faced a lower risk of ischemic stroke than patients…
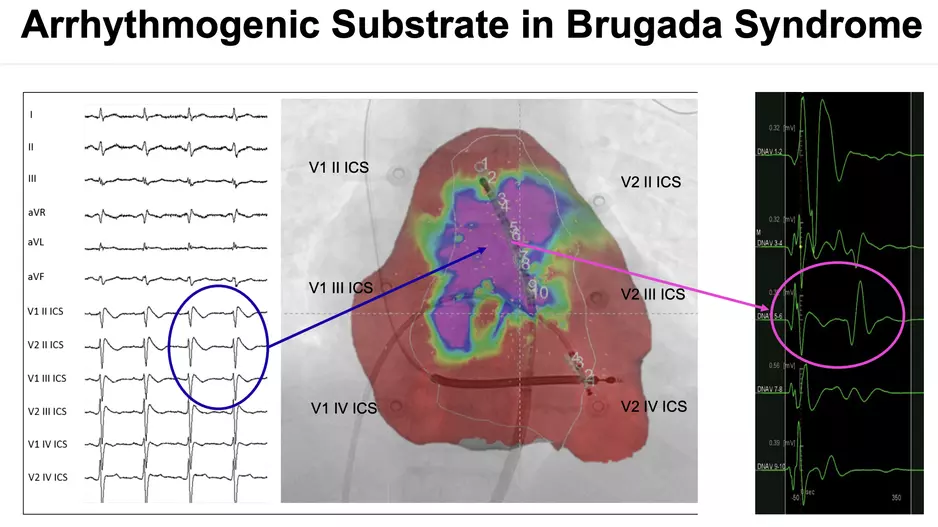
A late-breaking study found epicardial ablation in Brugada syndrome (BrS) helped significantly reduce sudden cardiac arrest in these patients, increasing…
A large proportion of patients with atrial fibrillation (AFib) end up in emergency rooms (ERs), but there are inherent issues with most hospital ERs handing off these patients to…
The Short-AF Study data presented as a late-breaking study at Heart Rhythm 2022 found a 96% percent freedom from reoccurrence of atrial…
Cardiologist Morton M. Mower, MD, co-inventor of the automatic implantable cardioverter defibrillator (ICD), has died of cancer. He was 89 years old.
The Baltimore Sun published a heartfelt tribute…
Same-day discharge (SDD) is an effective treatment option for patients undergoing atrial fibrillation (AFib) ablation, according to new findings presented at Heart Rhythm 2022. …
Here is a list with links for all of the late-breaking studies presented at the Heart Rhythm Society (HRS) 2022…
The U.S. Food and Drug Administration (FDA) has approved mavacamten, sold by Bristol Myers Squibb (BMS) under the name Camzyos, for the treatment of adult patients…
New data from the STROKE AF clinical trial, which showed that large and small vessel ischemic stroke patients who receive short-term or intermittent monitoring with Holter and 30-day…
Same-day discharge (SDD) after left atrial appendage occlusion (LAAO) is a safe treatment option that does not lead to worse patient outcomes, according to new findings published in the American Journal of Cardiology.[1]…
Using drones to deliver automated external defibrillators (AEDs) is an effective way to reach patients suffering from out-of-hospital cardiac arrest (OHCA), according to a new analysis published in European Heart Journal.[1…
Fitbit’s new algorithm for identifying signs of atrial fibrillation (AFib) has been cleared by the FDA.
The photoplethysmography (PPG) algorithm was designed to passively evaluate a user’s heart…
Among patients who received a smartwatch notification that they may have atrial fibrillation (AFib), 94% of those who followed up with a doctor were confirmed to indeed have the common…
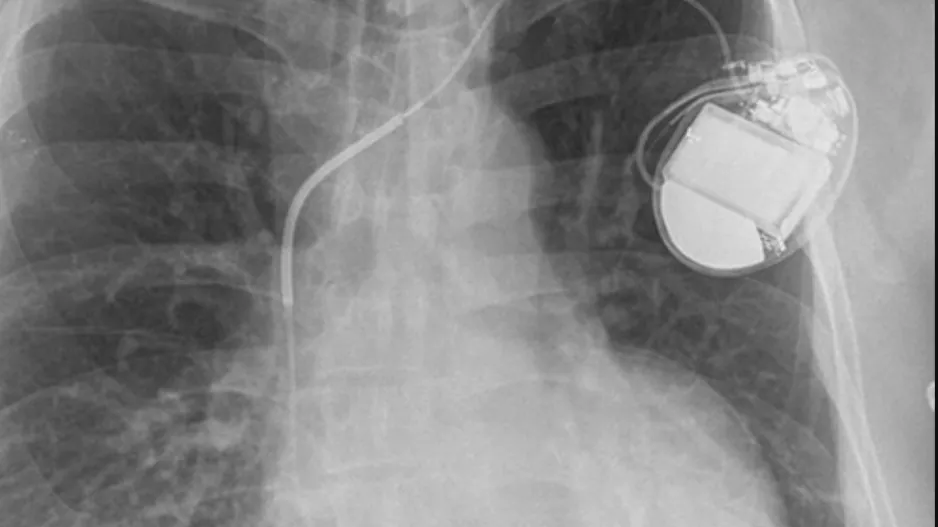
The U.S. Food and Drug Administration (FDA) has approved Abbott’s Aveir single-chamber (VR) leadless pacemaker as a treatment option for patients with slow heart rhythms. It is the second VR leadless pacemaker to gain approval in the…