More Articles
Empagliflozin, an SGLT2 inhibitor approved by the U.S. Food and Drug Administration for the treatment of type 2 diabetes and…
Teva Pharmaceuticals USA, a New Jersey-based healthcare company focused on the development and distribution of generic medications, has recalled a single lot of its …
Interventional cardiologists and other heart specialists from all over the world traveled to Atlanta this week for SCAI 2022, the 45th annual meeting of the Society for Cardiovascular Angiography and…
Intravascular lithotripsy (IVL), the FDA-cleared technology developed by Shockwave Medical, is associated with better peripheral artery disease (PAD) outcomes after two years than…
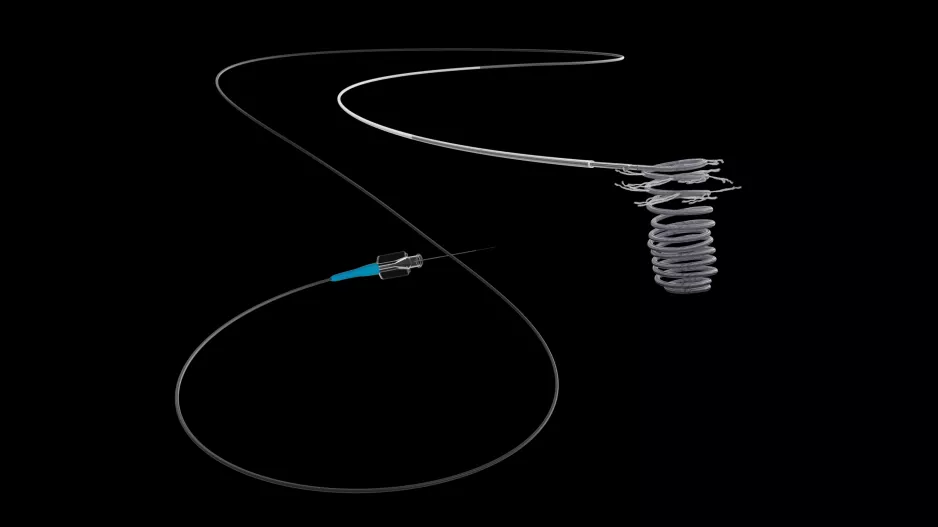
In a Series D financing round, Ireland-based vessel closure device company Vivasure raised $23 million to advance clinical development and regulatory approval for its absorbable, patch-based, percutaneous…
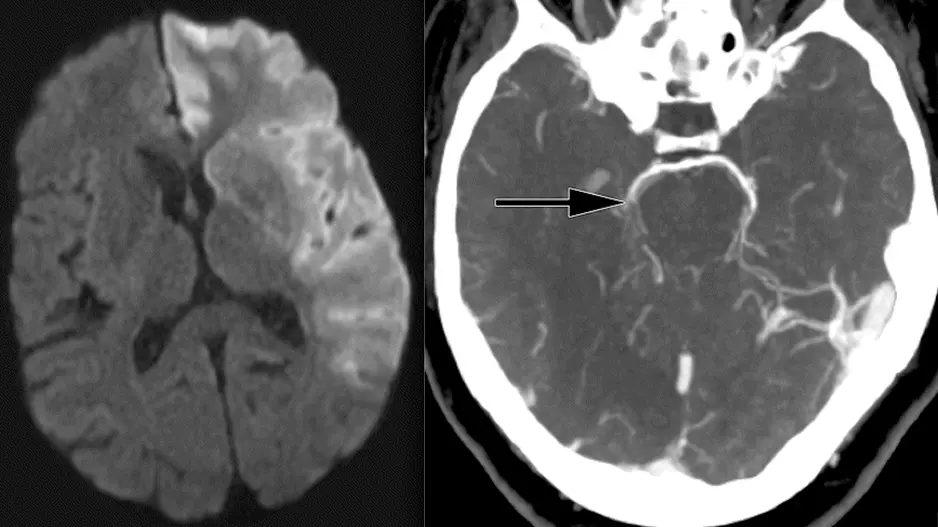
Certain strategies for treating or preventing intracerebral hemorrhages (ICHs), also known as bleeding strokes, may not be as effective as physicians previously believed, according to a new guidance document from the…
Improved shielding can lead help limit radiation exposure among endovascular aortic repair (EVAR) operators, according to a new analysis published in the European Journal of Vascular & Endovascular Surgery.[1]…
A team of cardiologists and surgeons at Hackensack University Medical Center in New Jersey believe they have made history by…
Carotid endarterectomy (CEA) is not associated with a significant reduction in the risk of dementia, according to a new analysis…
It has been a busy few weeks for the U.S. Food and Drug Administration (FDA). The agency…
Introducing hospitals to different decision support tools (DSTs) can improve the level of shared decision-making (SDM) among vascular surgeons and their patients, according to a new study published in the European Journal of…
Acknowledging the complexity and multifaceted nature of caring for patients with chronic limb-threatening ischemia (CLTI), eight medical societies cooperated to develop and release a …
The U.S. Preventive Services Task Force (USPSTF) has published its final recommendation statement on the practice of taking daily low-dose aspirin to prevent…
Transit Scientific, a Salt Lake City-based healthcare technology company, has received U.S. FDA clearance for its XO Cross Support Catheter Platform, already approved for peripheral vascular…
A history of allergic disorders — including asthma, digestive allergies and skin allergies — is associated with a heightened risk of high blood pressure and/or coronary heart disease, according to a new analysis of more than 34,000…
A new study of the U.S. Department of Veterans Affairs Veterans Health Administration (VHA) National Telestroke Program by the Regenstrief Institute and Indiana University School of Medicine shows the program prevents unnecessary hospital…
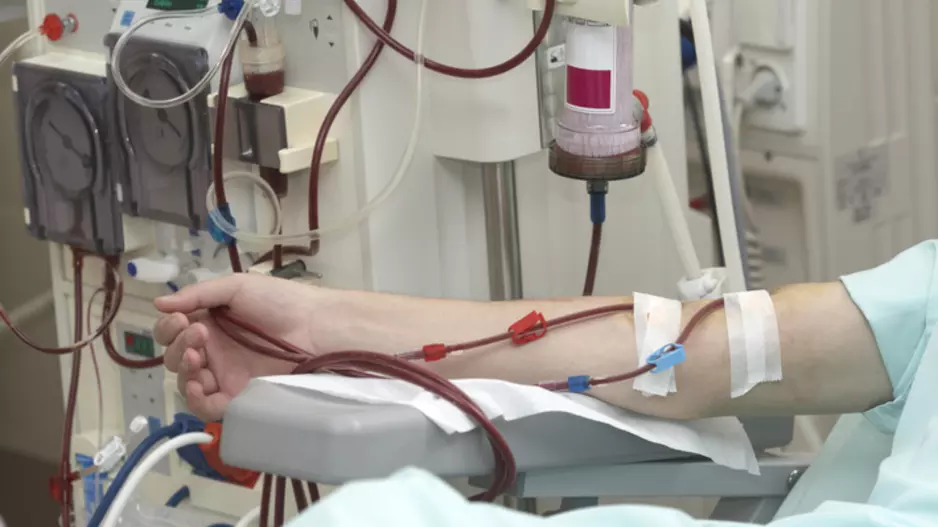
Mortality rates from myocardial infarction (MI), stroke and pulmonary embolism (PE) improved from 1998 to 2015 among patients receiving dialysis, according to new findings published in JAMA Network Open.[1] In fact, mortality…
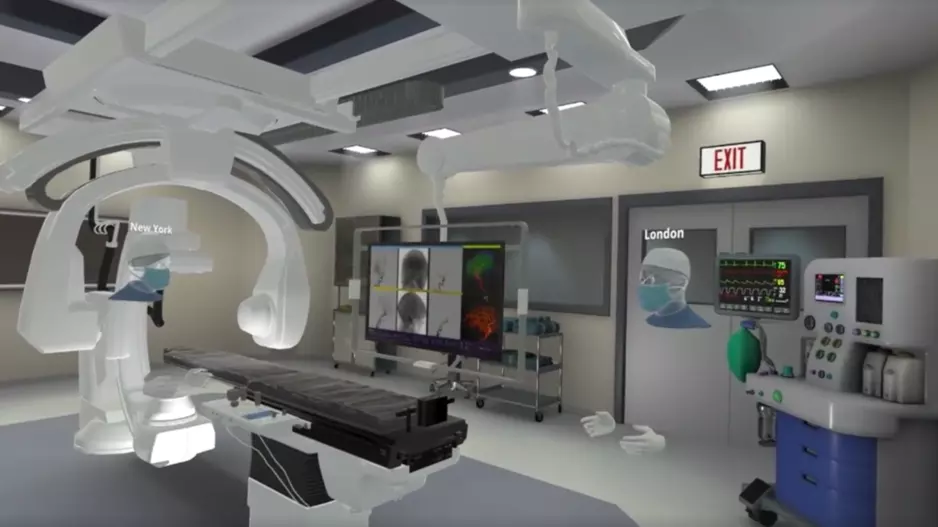
FundamentalVR, a London-based healthcare technology company that designs virtual reality (VR) solutions, has announced that it will now be offering haptic training simulations focused on…
When treating patients at a high bleeding risk (HBR) with percutaneous coronary intervention (PCI), the use of a drug-coated balloon (DCB) appears to be as safe and effective as a current-generation drug-eluting stent (DES), according…
Certain low-risk ST-segment elevation myocardial infarction (STEMI) patients can undergo percutaneous coronary intervention (PCI) without stenting, according to new findings published in EuroIntervention.[1]…
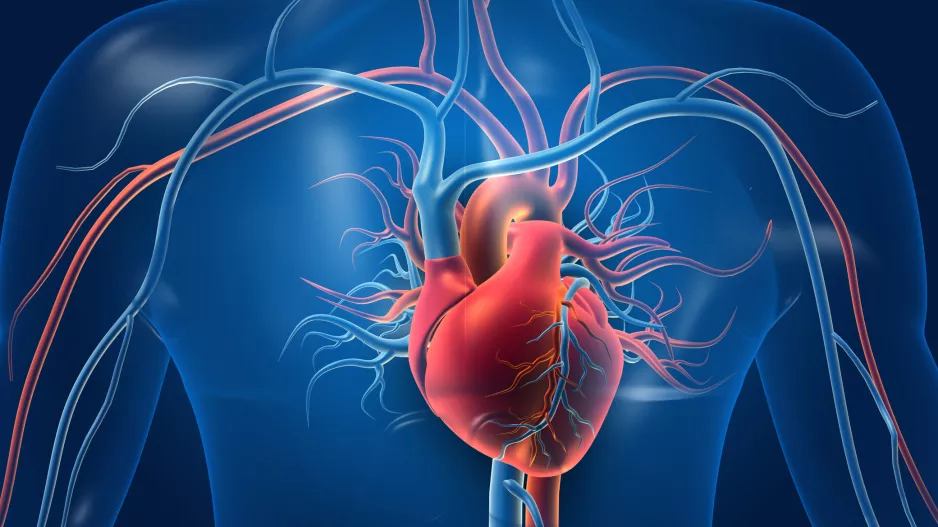





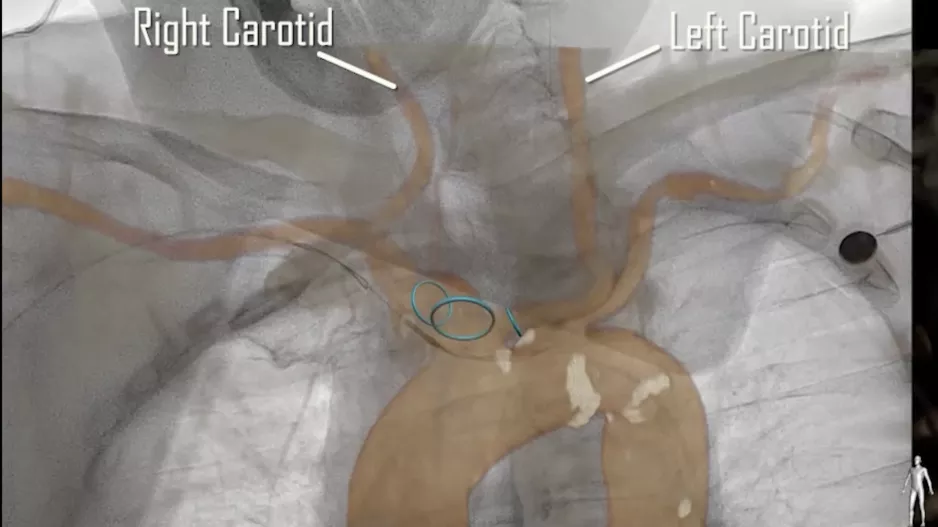

![Carotid endarterectomy (CEA) is not associated with a significant reduction in the risk of dementia, according to a new analysis published in the European Journal of Vascular & Endovascular Surgery.[1] However, the study’s authors noted, it did still lead to considerable improvements in stroke risk.](/sites/default/files/styles/media_image/public/2022-05/2006-07-12-1-carotidartery.jpg.webp?itok=iGTcsRHl)