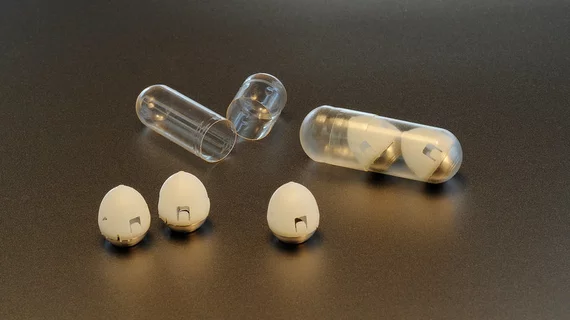
A capsule no bigger than a blueberry could be the future of insulin delivery for type 1 diabetics, according to researchers at MIT.
Giovanni Traverso, PhD, an assistant professor at Brigham and Women’s Hospital at Harvard Medical School and a visiting scientist in MIT’s department of mechanical engineering, co-led the group of scientists who developed the pill, a biodegradable capsule that contains a small needle made of compressed insulin. The insulin self-injects once it reaches a patient’s stomach, circumventing the need for uncomfortable daily injections.
“Our motivation is to make it easier for patients to take medication, particularly medications that require an injection,” Traverso said in a press release. “The classic one is insulin, but there are many others.”
Any protein drugs that typically need to be injected, like the immunosuppressants used to treat rheumatoid arthritis or inflammatory bowel disease, could benefit from this new method, he said. It might even work for nucleic acids like DNA and RNA.
The innovation stems from one of Traverso and his colleagues’ past studies, in which they developed a pill coated with tiny needles to inject drugs into the lining of the small intestine or stomach. But that design wasn’t streamlined and allowed for the possibility of drugs to inject themselves into the interior of the stomach, where they would be broken down by stomach acids before they could have any effect.
The researchers pared their design down to a capsule fitted with a singular needle—one made of nearly 100 percent compressed, freeze-dried insulin. The shaft of the needle is made from a biodegradable material and the needle itself is attached to a compressed spring held in place by a disk made of sugar. When a patient swallows the capsule, water in the stomach dissolves the sugar disk, releasing the spring and injecting the needle into the stomach wall.
Traverso said his colleagues assume diabetic patients won’t be able to feel the injection, since the stomach wall has no pain receptors. The method hasn’t been tested in humans yet, but the researchers recently wrapped a study in which they successfully delivered 300 micrograms of insulin to pig models using the capsule approach. Even more recently, Traverso said they’ve been able to increase that dose to 5 milligrams, a comparable amount to that a type 1 diabetic would need to take.
To ensure the capsule injects its needle properly, the MIT team made the pill bottom-heavy, drawing inspiration from the African leopard tortoise, whose steep shell allows it to right itself if it rolls onto its back. So, even in the volatile environment of the stomach, the capsule will reorient itself for a successful injection.
“As soon as you take it, you want the system to self-right so that you can ensure contact with the tissue,” Traverso said.
Once the tip of the needle is injected into the stomach wall, the insulin dissolves at a pace set by the research team. In the pig study, it took about an hour for all the insulin to release into the animals’ bloodstreams.
“Initial tests on animals shows insulin delivered in this manner controls blood sugar as well as a normal injection,” Traverso and his colleagues wrote in Science, where their latest findings were published Feb. 8. “Along with other related advances in the field, the new approach holds out new hope that patients may soon be able to do away with needles.”