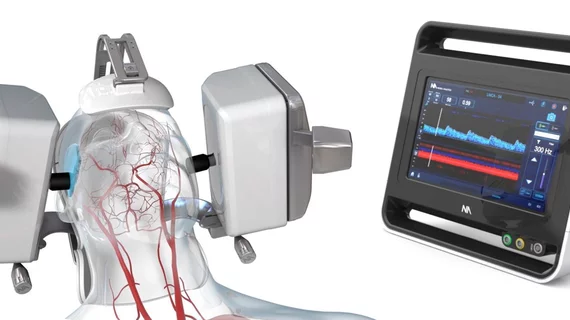
The FDA has cleared a robotic system that can noninvasively assess abnormal blood flow to the brain, helping diagnose and triage patients with ischemic strokes and other neurologic disorders.
The NeuralBot system, manufactured by Neural Analytics, automatically adjusts the orientation and position of its ultrasound products to help monitor a patient’s blood flow, according to a press release announcing the FDA approval. It was approved after research showed the system collected blood flow data similar to that collected manually by five expert technicians using traditional ultrasound.
“Progress in treating neurological disorders has lagged due to a lack of available low-cost and objective patient diagnostic information,” said Robert Hamilton, PhD, cofounder and Chief Scientific Office of Neural Analytics. “This has resulted in misdiagnosis, treatment delays and additional healthcare expenditures for patients suffering neurological disease. Our new technology can assist a healthcare professional—when an expert technician is not available—with the collection of blood flow data to assist clinicians in efficiently triaging patients for appropriate treatment.”
Neural Analytics already received FDA approval for its Lucid Robotic System, which contains the ultrasound equipment utilized by the NeuralBot technology. The company plans to immediately commercialize the two products as a package deal, according to the release.